Coral Polyp Anatomy
Corals, often mistaken for plants due to their flower-like appearance, are animals that generally exist in the form of colonies. These colonies attach to oceanic substrate and when numerous colonies form a consortium, a coral reef arises. Corals are members of the Animal Kingdom with classification in the Phylum Cnidaria; further classification places them in the class Anthozoa that is closely related to the class Scyphyzoa (e.g. jelly fishes) and the class Hydrozoa (e.g. Hydra).
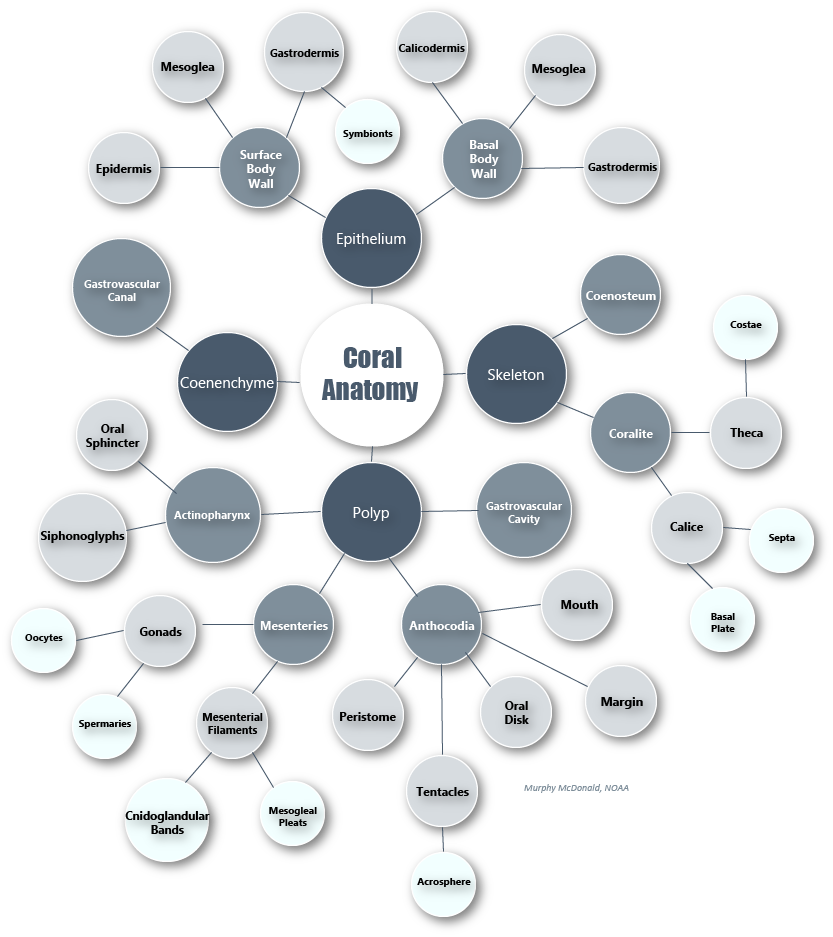
The polyp is the basic living unit of Anthozoa (further subdivided into subclass Hexacorallia, which includes stony corals, and subclass Octocorallia, which includes soft corals). Polyps can occur either singularly, as in the case of anemones and some stony corals, or can form colonies, as they do in most stony and all soft corals. A key feature of Cnidaria is the presence of cnidocytes, or stinging cells, which the polyp uses for prey capture and defense.



Polyp Anatomy
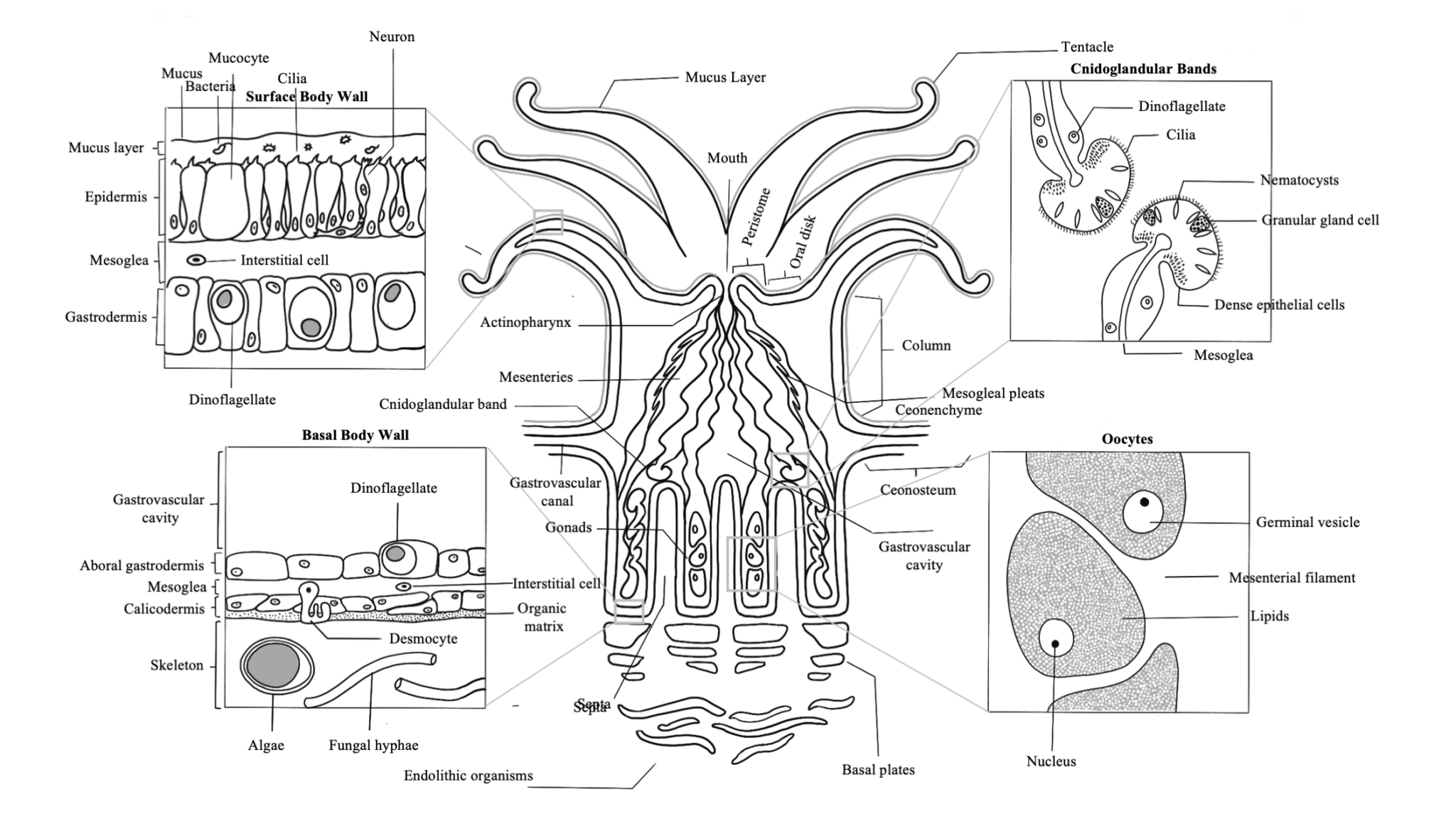
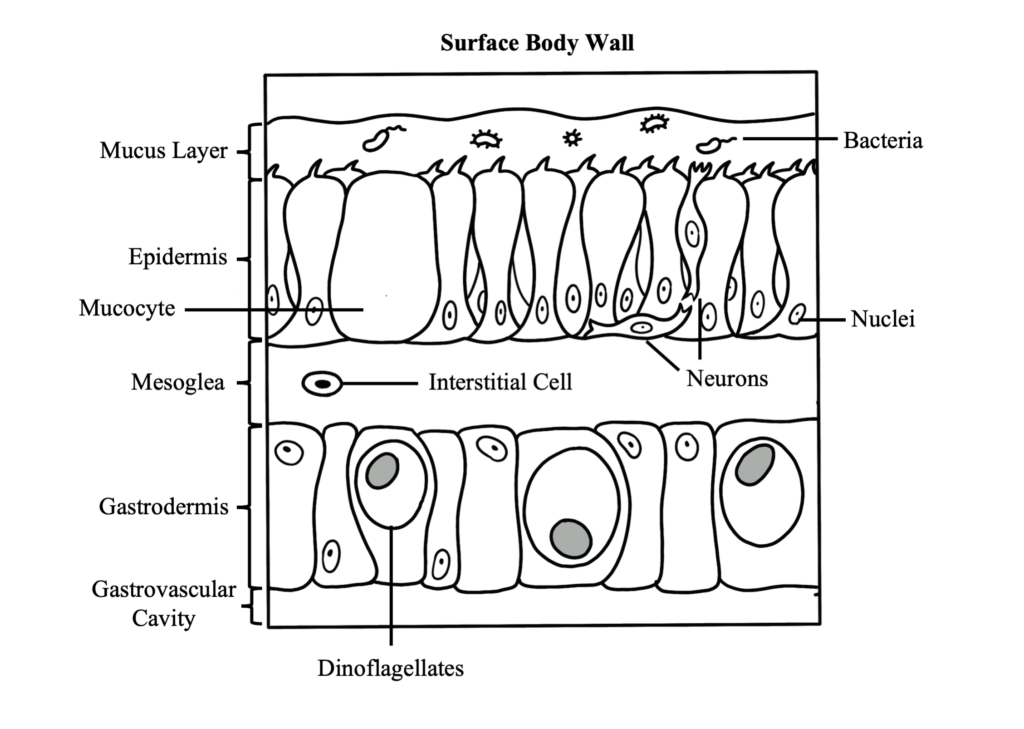
The fundamental structure of a coral polyp is a cylindrical sac composed of two layers of epithelia: the surface body wall, which forms the outermost barrier between the organism and its surroundings, and the basal body wall, which anchors the polyp to its skeleton. Water and ingested materials enter the polyp through the mouth at the distal (uppermost) region of the polyp; water and waste products are expelled by the polyp through the same opening. Respiration and excretion occur by direct exchange of molecules through the two layers of epithelia.
The portion of the polyp that is able to extend beyond the skeleton is called the column. Located at the distal end of this region is the polyp’s mouth, oral disk, and ring of tentacles. The mouth is surrounded by the peristome, which is an elevated region surrounding the mouth opening, and the oral disk, which extends from the peristome to a ring of tentacles around its perimeter. Tentacles are studded with stinging cells called nematocysts, and in some cases, can be topped with a bulbous structure called an acrosphere.
Tentacles are used by the coral polyp to capture and ingest particulate and planktonic food from the environment. For corals that live within the photic zone (the depths where light penetrates into the ocean) the nutrients that the coral acquires through this heterotrophic method supplements those derived from the algal symbionts that live within its internal tissue layer, known as the gastrodermis. The symbionts that live within corals are microscopic dinoflagellates of the family Symbiodinacea, which undergo photosynthesis and exchange nutrients and waste molecules with the coral host cells. This consortium of algae within coral tissues contributes to their color and drives the coral’s need for light in shallow reef habitats.

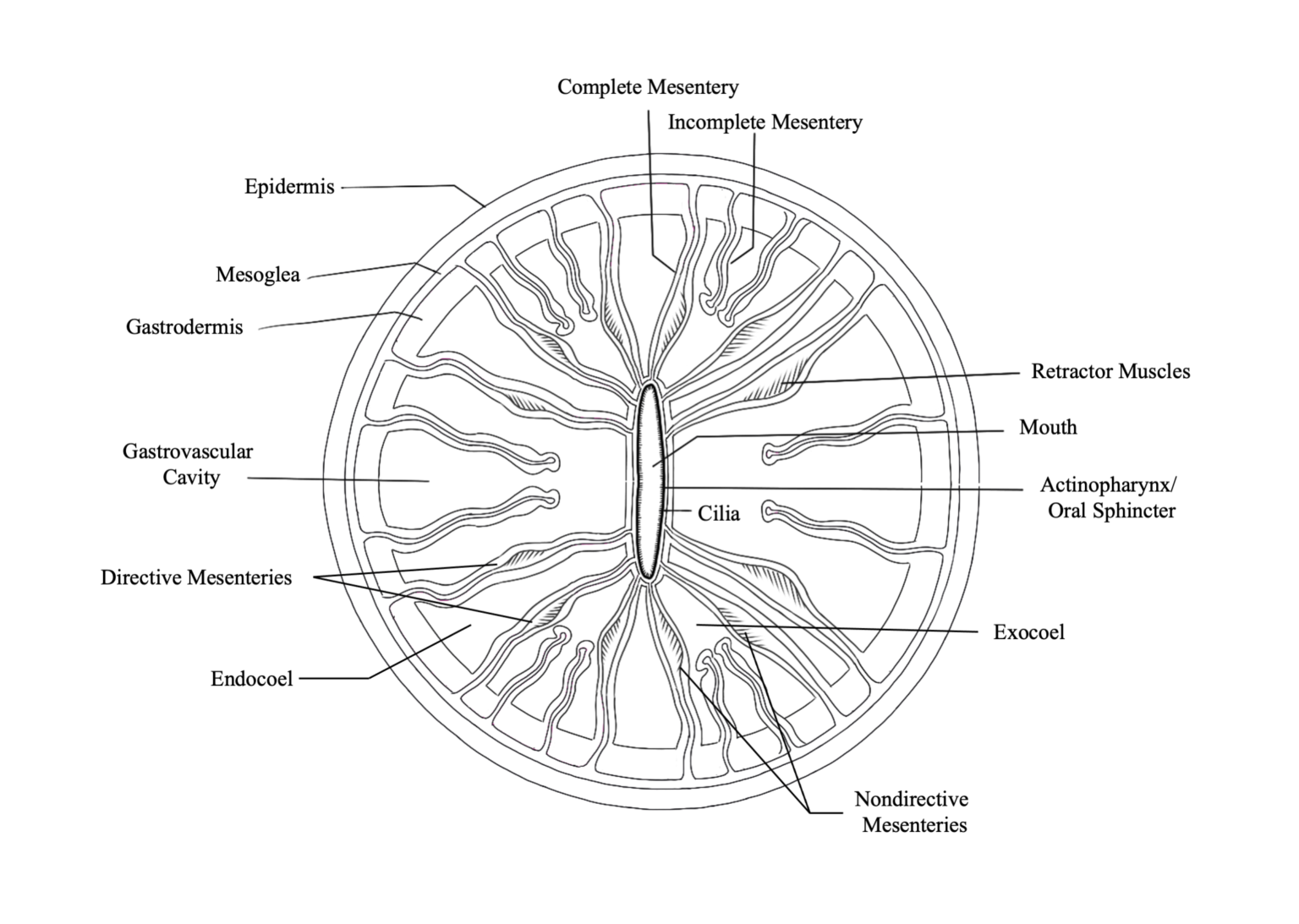
A defining feature of anthozoans is the actinopharynx, which is an invagination of the epidermis that forms a short muscular passageway from the mouth to the gastric cavity. The actinopharynx controls the opening and closing of the polyp mouth by using a ring of muscle fibers called the oral sphincter that is located on its distal edge. The lumen of the actinopharynx is lined with epithelial cells with elongated cilia to move material in and out of the polyp mouth.
Extending down from the actinopharynx into the interior cavity of the polyp are tissue partitions known as mesenteries. Mesenteries provide structural support to the polyp column and are also important for digestion, as they increase the surface area of the interior epithelia for gas exchange and nutrient absorption. Retractor muscles on mesenteries near the body wall allow the polyp to contract and extend. Mesenteries that are attached to the actinopharynx are known as “complete” or “perfect” mesenteries, those that are not connected are known as “incomplete” or “imperfect.” The open space within the polyp between mesenteries is known as the gastrovascular cavity.
Mesenterial filaments are the thickened and elongated free edges of mesenteries below the actinopharynx. These can be stretched out of the mouth to help capture and digest food outside and inside of the polyp using their high abundance of granulated gland cells, which secrete digestive enzymes. Bulbous lobes at the end of mesenterial filaments are called cnidoglandular bands. These structures contain a battery of nematocysts for catching prey and for defense, and of granular gland cells to aid in digestion.
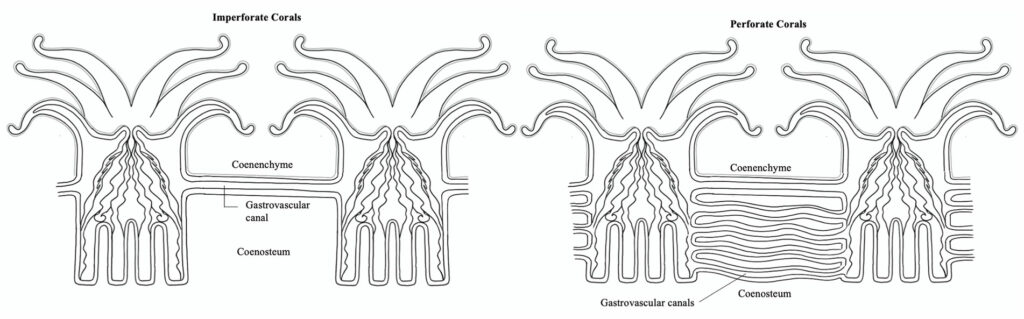
In coral colonies, the common tissue that surrounds and links polyps is known as the coenenchyme. Within the coenenchyme, tubes called gastrovascular canals link the gastrovascular cavities of polyps throughout the colony. This network of tubes is what allows individual polyps to act as a unified organism by shuttling resources and cellular signals throughout the colony. In imperforate corals, polyps of a colony are separated by solid skeleton (known as the coenosteum), so this network of canals runs just under the surface of the coenenchyme above the skeleton. In perforate corals, numerous gastrovascular canals pierce the more porous skeleton between polyps
Learn More about the Essentials of Coral Biology:
Skeletal Anatomy
Learn about all of the components of the coral skeleton and the different morphologies that it can display
Coral Reproduction
Learn about how corals use different reproductive modes to overcome their immobility
Publications Relevant to Coral Anatomy
Agassiz, L. (1849) On the structure of coral animals. Proceedings of the American Association for the Advancement of Sciences 2, 68-77.
Brown, B. E., & Bythell, J. C. (2005). Perspectives on mucus secretion in reef corals. In Marine Ecology Progress Series (Vol. 296). https://doi.org/10.3354/meps296291
Burton, P. M. (2008). Insights from diploblasts; the evolution of mesoderm and muscle. In Journal of Experimental Zoology Part B: Molecular and Developmental Evolution (Vol. 310, Issue 1). https://doi.org/10.1002/jez.b.21150
Cairns, S. D. (1991). A revision of the Ahermatypic Scleractinia of the Galapagos and Cocos Islands. Smithsonian Contributions to Zoology, 504. https://doi.org/10.5479/si.00810282.504
Chornesky, E., and Peters, E. (1987) Sexual Reproduction and colony growth in the reef coral Porites astreoides. Biological Bulletin 172, 161-177.
Epp, L., Smid, I., & Tardent, P. (1986). Synthesis of the mesoglea by ectoderm and endoderm in reassembled hydra. Journal of Morphology, 189(3). https://doi.org/10.1002/jmor.1051890306
Fautin, D., and Mariscal, R. (1991) Cnidaria: Anthozoa, in Microscopic Anatomy of Invertebrates: Placozoa, Porifera, Cnidaria, and Ctenophora (eds F. harrison and J. Westfall), Vol. 2. Wiley-Liss, New York, NY, pp. 267-358
Galloway, S., Work, T., Bochsler, V., et al. (2007) Coral Disease and Health Workshop: Coral Histopathology II. NOAA Technical Memorandum NOS NCCOS 56 and CRCP 4. National Oceanic and Atmospheric Administration, Silver Spring, MD.
Gateño, D., & Rinkevich, B. (2003). Coral polyp budding is probably promoted by a canalized ratio of two morphometric fields. Marine Biology, 142(5). https://doi.org/10.1007/s00227-003-1009-8
Gladfelter, E. H. (1932) Circulation of fluids in the gastrovascular system of the reef coral Acropora cervicornis. The Biological Bulletin, 165(3). https://doi.org/10.2307/1541469
Goldberg, W. M. (2002). Feeding behavior, epidermal structure and mucus cytochemistry of the scleractinian Mycetophyllia reesi, a coral without tentacles. Tissue and Cell, 34(4). https://doi.org/10.1016/S0040-8166(02)00009-5
Goldberg, W. M. (2002). Gastrodermal structure and feeding responses in the scleractinian Mycetophyllia reesi, a coral with novel digestive filaments. Tissue and Cell, 34(4). https://doi.org/10.1016/S0040-8166(02)00008-3
Goreau, T. F. (1956) A study of the Biology and Histochemistry of Corals. Ph. D. thesis, Yale University.
Grimmelikhuijzen, C. J., & Westfall, J. A. (1995). The nervous systems of cnidarians. In EXS (Vol. 72). https://doi.org/10.1007/978-3-0348-9219-3_2
Lang, J. (1984) Whatever works: The variabie importance of skeletal and of non-skeletal shcaracters in scleractinian taxonomy. Paleontographica Americana 54, 18-44.
Lang, J. C., & Chornesky, E. (1990). Competition between scleractinian reef corals – a review of mechanisms and effects. In Ecosystems of the world. Volume 25: Coral reefs (Vol. 25).
Lesser, M. P., Mazel, C. H., Gorbunov, M. Y., & Falkowski, P. G. (2004). Discovery of symbiotic nitrogen-fixing cyanobacteria in corals. Science, 305(5686). https://doi.org/10.1126/science.1099128
Marshall, A. T., & Wright, O. P. (1993). Confocal laser scanning light microscopy of the extra-thecal epithelia of undecalcified scleractinian corals. Cell & Tissue Research, 272(3). https://doi.org/10.1007/BF00318560
Martindale, M. Q., Pang, K., & Finnerty, J. R. (2004). Investigating the origins of triplosblasty: “Mesodermal” gene expression in a diploblastic animal, the sea anemone Nematostella vectensis (phylum, Cnidaria; class, Anthozoa). Development, 131(10). https://doi.org/10.1242/dev.01119
Mullen, K., Peters, E., and harvell, C. (2004) Coral resistance to disease, in Coral Health and Disease (eds E. Rosenberg and Y. Loya). Springer-Verlag, Berlin, pp. 377-399.
Muscatine, L., Ferrier-Pagès, C., Blackburn, A., Gates, R. D., Baghdasarian, G., & Allemand, D. (1998). Cell-specific density of symbiotic dinoflagellates in tropical anthozoans. Coral Reefs, 17(4). https://doi.org/10.1007/s003380050133
Muscatine, L., Tambutte, E., & Allemand, D. (1997). Morphology of coral desmocytes, cells that anchor the calicoblastic epithelium to the skeleton. Coral Reefs, 16(4). https://doi.org/10.1007/s003380050075
Nothdurft, L. D., & Webb, G. E. (2009). Clypeotheca, a new skeletal structure in scleractinian corals: A potential stress indicator. Coral Reefs, 28(1). https://doi.org/10.1007/s00338-008-0439-7
Östman, C. (2000). A guideline to nematocyst nomenclature and classification, and some notes on the systematic value of nematocysts. Scientia Marina, 64(SUPPLEMENT 1). https://doi.org/10.3989/scimar.2000.64s131
Palmer, C. v., Bythell, J. C., & Willis, B. L. (2010). Levels of immunity parameters underpin bleaching and disease susceptibility of reef corals. The FASEB Journal, 24(6). https://doi.org/10.1096/fj.09-152447
Palmer, C. v., Modi, C. K., & Mydlarz, L. D. (2009). Coral fluorescent proteins as antioxidants. PLoS ONE, 4(10). https://doi.org/10.1371/journal.pone.0007298
Palmer, C. v., Mydlarz, L. D., & Willis, B. L. (2008). Evidence of an inflammatory-like response in non-normally pigmented tissues of two scleractinian corals. Proceedings of the Royal Society B: Biological Sciences, 275(1652). https://doi.org/10.1098/rspb.2008.0335
Peters, E., (1984) A Survey of the Normal and Pathological Histology of Scleractinian Corals with Emphasis on the Effects of Sedimentation Stress. Ph. D. dissertation, University of Rhode Island.
Peters, E., Price, K., & J. Borsay Horowitz, D. (2005). Histological preparation of invertebrates for evaluating contaminant effects. In Techniques in Aquatic Toxicology, Volume 2. https://doi.org/10.1201/9780203501597.ch36
Petes, L. E., Harvell, C. D., Peters, E. C., Webb, M. A. H., & Mullen, K. M. (2003). Pathogens compromise reproduction and induce melanization in Caribbean sea fans. Marine Ecology Progress Series, 264. https://doi.org/10.3354/meps264167
Puverel, S., Tambutté, E., Zoccola, D., Domart-Coulon, I., Bouchot, A., Lotto, S., Allemand, D., & Tambutté, S. (2005). Antibodies against the organic matrix in scleractinians: A new tool to study coral biomineralization. Coral Reefs, 24(1). https://doi.org/10.1007/s00338-004-0456-0
Seipel, K., & Schmid, V. (2006). Mesodermal anatomies in cnidarian polyps and medusae. In International Journal of Developmental Biology (Vol. 50, Issue 7). https://doi.org/10.1387/ijdb.062150ks
Sharp, K. H., Ritchie, K. B., Schupp, P. J., Ritson-Williams, R., & Paul, V. J. (2010). Bacterial acquisition in juveniles of several broadcast spawning coral species. PLoS ONE, 5(5). https://doi.org/10.1371/journal.pone.0010898
Vargas-Ángel, B., Peters, E. C., Kramarsky-Winter, E., Gilliam, D. S., & Dodge, R. E. (2007). Cellular reactions to sedimentation and temperature stress in the Caribbean coral Montastraea cavernosa. Journal of Invertebrate Pathology, 95(2). https://doi.org/10.1016/j.jip.2007.01.003
Veron, J. (2000) Corals of the World, Volume 1, Australian Institute of Marine Science, Townsville, Queensland, Australia.
Zilberberg, C., & Edmunds, P. J. (1999). Patterns of skeletal structure variability in clones of the reef coral Montastraea franksi. Bulletin of Marine Science, 64(2).
Zilberg, C., and Edmunds, P. (1999) Patterns of skeletal structure variability in clones of the reef coral Montastrea franksii. Bulletin of Marine Science, 64, 373-381
 Official websites use .gov
A .gov website belongs to an official government organization in the United States.
Official websites use .gov
A .gov website belongs to an official government organization in the United States. Secure .gov websites use HTTPS
A lock or https:// means you’ve safely connected to the .gov website. Share sensitive information only on official, secure websites.
Secure .gov websites use HTTPS
A lock or https:// means you’ve safely connected to the .gov website. Share sensitive information only on official, secure websites.